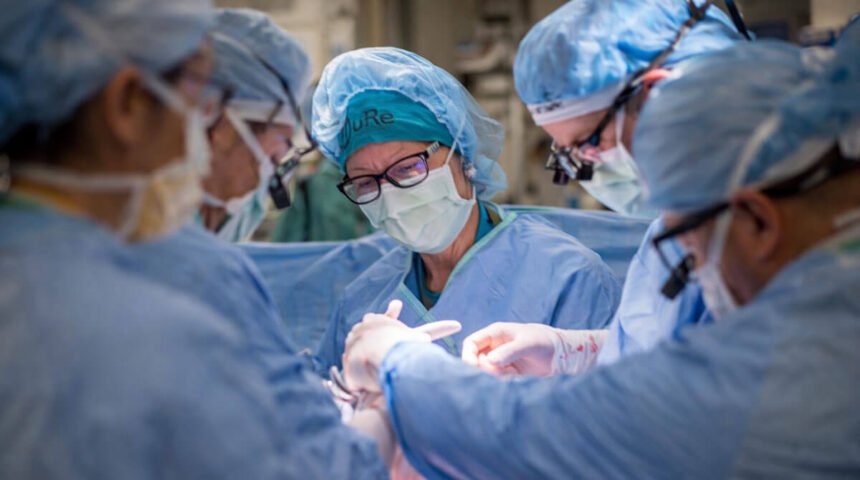
In February 2026, a team of surgeons at UC Davis Health in California quietly changed the history of medicine. They published results in The Lancet — one of the most respected medical journals in the world — showing they had safely performed the world’s first in-utero stem cell therapy for spina bifida. Six unborn babies had their exposed, damaged spinal cords treated with living stem cells while still in the womb. Every single one was born with their repair intact. Not one required emergency brain intervention. And at least one child is now four years old — walking, running, and jumping.
- What Is Spina Bifida? Understanding the Condition Behind the Breakthrough
- The Road to a Breakthrough: 25 Years of Fetal Surgery Research
- How the In-Utero Stem Cell Therapy for Spina Bifida Actually Works
- The Results: What The Lancet Study Found in 2026
- Why This Spina Bifida Breakthrough Is Bigger Than One Disease
- What This Means for Cambodia and Southeast Asia
- Progress Is Real — and the World Is Getting Better
- Frequently Asked Questions: Spina Bifida Treatment Before Birth
- What You Can Do
This is the story of how they did it, why it matters to families everywhere — including in Southeast Asia and Cambodia — and what it signals about the future of medicine for children not yet born.
What Is Spina Bifida? Understanding the Condition Behind the Breakthrough
What Happens When the Spinal Cord Does Not Close
Spina bifida — known medically as myelomeningocele in its most severe form — is a congenital birth defect caused by the incomplete closure of the neural tube during the first 28 days of pregnancy. In babies with spina bifida, this process fails. Part of the spinal cord is left exposed, protruding through a gap in the spine, unprotected by bone or skin.
The consequences are serious and lifelong: partial or full paralysis from the waist down, loss of bladder and bowel control, a brain complication called hindbrain herniation, hydrocephalus requiring a surgically implanted shunt, and in some cases learning difficulties and reduced life expectancy.
How Common Is Spina Bifida — Globally and in Southeast Asia?
Worldwide, spina bifida and related neural tube defects affect approximately 300,000 newborns every year. In Southeast Asia the burden is higher, at an estimated 13 per 10,000 live births. In Cambodia and many neighbouring countries, mandatory folic acid fortification of staple foods has not been fully implemented — meaning preventable cases continue to occur at rates that policy change could dramatically reduce.
The Road to a Breakthrough: 25 Years of Fetal Surgery Research
The MOMS Trial: How Fetal Surgery First Proved Its Value
The CuRe Trial’s roots trace back to a landmark 2011 study in The New England Journal of Medicine — the Management of Myelomeningocele Study (MOMS), co-led by Diana Farmer. The MOMS trial proved that prenatal surgery produced dramatically better outcomes than postnatal surgery: reduced need for brain shunting, reversal of hindbrain herniation, and improved motor outcomes. The trial was stopped early because the benefits were already so clear it would have been unethical to continue the control group. Fetal surgery for spina bifida became the new standard of care.
But Diana Farmer was not satisfied. Even children who underwent fetal surgery were, in many cases, still requiring wheelchairs. The surgery was helping. It was not healing.
Enter the CuRe Trial: Could Stem Cells Do What Surgery Could Not?
Farmer recruited Aijun Wang, a biomedical engineer, to take the research further. Together they built the UC Davis Health Surgical Bioengineering Laboratory and spent over a decade testing their approach in animal models. Their CuRe Trial — Cellular Therapy for In Utero Repair of Myelomeningocele — launched in the spring of 2021 as the world’s first attempt to combine fetal surgery with a live stem cell patch in a human patient.
“I’ve been working toward this day for almost 25 years.” — Diana Farmer, Principal Investigator, CuRe Trial
How the In-Utero Stem Cell Therapy for Spina Bifida Actually Works
The Stem Cell Patch: From Donated Placentas to the Operating Room
The stem cells used are placenta-derived mesenchymal stromal cells (PMSCs) — harvested from donated placental tissue. Wang’s engineering team developed a biodegradable scaffold seeded with the cells at 300,000 cells per square centimetre, creating the stem cell patch. Manufacturing takes four days and must be precisely timed for cell viability at surgery.
The Surgery, Step by Step
A team of around 40 specialists prepares. The mother is placed under general anaesthetic. A small opening is made in the uterus. The fetus is positioned to expose the affected spine. Standard fetal repair closes the spinal opening. Then — in the step that had never been performed in human history — the stem cell patch is applied directly onto the fetal spinal cord. The uterus is closed. The pregnancy continues.
Who Were the First Patients?
The Phase 1 trial enrolled six pregnant women between 24 and 26 weeks of gestation whose babies had been diagnosed with myelomeningocele. The first patient was Emily from Austin, Texas, whose daughter Robbie became the first human to receive this treatment on July 12, 2021. Michelle and Jeff from Oregon enrolled their son Tobi as the second patient. Each family relocated temporarily to Sacramento — accepting risks never before quantified in a human being.
The Results: What The Lancet Study Found in 2026
Complete Safety Record Across All Six Patients
The Phase 1 results published in The Lancet confirmed the treatment is safe. All six surgeries were successfully completed. Zero infections. Zero spinal fluid leaks. Zero abnormal tissue growth or tumours. Every wound healed completely. No mother or baby experienced serious adverse events attributable to the stem cells.
Brain Complications Reversed in Every Case
Post-birth MRI scans confirmed hindbrain herniation — the dangerous downward displacement of the lower brain — was reversed in all six babies. This is considered a key marker of surgical success, and its consistent achievement across all patients is highly significant.
No Brain Shunts Required at Discharge
Not one of the six babies required a brain shunt upon discharge — a common necessity in standard fetal surgery. This suggests the stem cells are providing neurological protection beyond what surgery alone can achieve.
“This is a major step toward a new kind of fetal therapy — one that doesn’t just repair but potentially helps heal and protect the developing spinal cord.” — Aijun Wang, Co-Principal Investigator
A Four-Year-Old Walking, Running, and Jumping
Tobi, the second patient, is now four years old — walking, running, and jumping, with no wheelchair and no bladder interventions needed. His mother Michelle said the decision to join the trial was life-changing.
Phase 2 Approved: Up to 35 Patients
Because Phase 1 results were so strong, both the FDA and an independent safety monitoring board approved the CuRe Trial to advance to Phase 2, which will enrol up to 35 patients. Track the ongoing trial at ClinicalTrials.gov (NCT04652908).

Why This Spina Bifida Breakthrough Is Bigger Than One Disease
This is a proof of concept for an entirely new category of medicine: prenatal regenerative therapy. Wang has already received a separate NIH grant to explore in-utero gene editing for Dup15q syndrome, a chromosomal disorder affecting brain development. Farmer has spoken about extending stem cell fetal therapy to congenital heart defects, diaphragmatic hernias, and other conditions managed only after birth.
The scientific community has responded with measured enthusiasm. Shine, the UK’s leading spina bifida and hydrocephalus charity, described the Phase 1 results as very encouraging and a major milestone — while noting that longer-term safety follow-up remains essential.
What This Means for Cambodia and Southeast Asia
The Folic Acid Gap — A Solvable Problem Right Now
For readers in Cambodia, this story is both hopeful and urgent. The most powerful tool we currently have against spina bifida is folic acid. According to the WHO and the CDC, folic acid taken daily before conception and through early pregnancy reduces neural tube defect risk by up to 70%. Many countries have mandated fortification of staple foods — and have seen spina bifida rates fall dramatically. Cambodia has not yet fully adopted this policy.
Cambodia’s Healthcare Infrastructure: Building Toward the Future
Advanced fetal surgery for spina bifida is largely unavailable in Southeast Asia today. But Cambodia is actively investing in its medical future. Superior Med King’s Hospital, set to become a landmark in the country’s healthcare landscape, signals that Cambodia is positioning itself as a regional hub for premium healthcare. As the country moves toward its ambitious economic milestones for 2030 and 2050, building the specialised surgical and neonatal infrastructure to deliver advanced fetal medicine must be part of the national vision.
A History Worth Honouring — and Building On
It is worth remembering that Cambodia has a proud medical heritage. The Khmer Empire built the world’s first organised healthcare system under King Jayavarman VII in the 12th century — hospitals with staff, management, and free care for all citizens. That spirit of universal care did not disappear; it lives on in Cambodia’s tradition of healing, and it is the spirit that modern medicine must be built upon.
Progress Is Real — and the World Is Getting Better
We live in an information environment that rewards alarm. Good news — especially the slow, scientific kind — rarely gets the headlines it deserves. But the world is, in measurable ways, getting better for the children born into it.
Child mortality is at its lowest point in recorded history. Vaccines have eliminated diseases that once killed millions. And now, for the first time, stem cells applied to the spinal cord of an unborn baby have been shown to be safe — and those children are walking.
At The Better Cambodia, we believe that telling these stories matters. Not because they erase the problems that remain — they don’t — but because they remind us that those problems are solvable. That is worth knowing. That is worth sharing. If you’d like to follow more global health breakthroughs and positive world news, explore our Health section.
Frequently Asked Questions: Spina Bifida Treatment Before Birth
Q: What is spina bifida treatment before birth?
A: In-utero spina bifida treatment involves operating on a baby’s exposed spinal cord while still in the womb, before 26 weeks of gestation. In 2026, the world’s first in-utero stem cell therapy for spina bifida was published in The Lancet, showing that applying a living stem cell patch during fetal surgery is safe — with strong results across all six trial patients.
Q: Can spina bifida be cured before birth?
A: Not yet fully cured, but the 2026 CuRe Trial results show in-utero stem cell therapy is safe and may significantly improve outcomes beyond surgery alone. All six babies were born with intact spinal repairs, reversed brain complications, and none required brain shunts. One child is now walking and running at age four.
Q: Who led the 2026 spina bifida stem cell trial?
A: The CuRe Trial was led by Diana Farmer and Aijun Wang at UC Davis Health, Sacramento, California. Farmer is the first woman ever appointed as a fetal surgeon and previously led the 2011 MOMS Trial that established in-utero surgery as the standard of care for spina bifida.
Q: How can spina bifida be prevented?
A: Taking folic acid (vitamin B9) daily before and during early pregnancy reduces neural tube defect risk by up to 70%, according to the WHO and CDC. Countries that have mandated folic acid fortification of staple foods have seen spina bifida rates fall dramatically. Cambodia and much of Southeast Asia have not yet fully implemented such programmes.
Q: Is spina bifida common in Cambodia and Southeast Asia?
A: Neural tube defects are more common in Southeast Asia than in countries with mandatory folic acid fortification. The regional prevalence is approximately 13 per 10,000 live births. In Cambodia, preventable cases continue to occur due to the absence of comprehensive food fortification policy — an area where advocacy can have immediate impact.
What You Can Do
If you are pregnant or planning to become pregnant, speak with your doctor about folic acid supplementation. Taking 400 micrograms daily before conception and through the first trimester is one of the most evidence-backed ways to reduce your baby’s risk of spina bifida.
If you work in public health or policy in Cambodia, folic acid fortification of staple foods deserves your attention. TBC Global Partners is actively working to bring healthcare investment into Cambodia — and voices from civil society that push for preventive health policy can shape that agenda.
And if you found this article valuable — share it. Stories like the CuRe Trial deserve wider attention. For more positive global health news and stories of progress from across the world and Cambodia, visit The Better Cambodia’s Health section.
Sources & References
All claims are sourced from peer-reviewed journals, academic medical institutions, and established science publications. Every source is live and verifiable.
| # | Citation & URL |
| [1] | Farmer DL, et al. Feasibility and safety of cellular therapy for in-utero repair of myelomeningocele (CuRe Trial): a first-in-human, phase 1, single-arm study. The Lancet. 2026;407(10531):867–875.https://doi.org/10.1016/S0140-6736(25)02466-3 |
| [2] | UC Davis Health. First-ever in-utero stem cell therapy for fetal spina bifida repair is safe, study finds. UC Davis Health News. February 26, 2026.https://health.ucdavis.edu/news/headlines/first-ever-in-utero-stem-cell-therapy-for-fetal-spina-bifida-repair-is-safe-study-finds/2026/02 |
| [3] | Nature News. World-first stem-cell therapy shows promise for treating spina bifida in the womb. Nature. February 26, 2026.https://www.nature.com/articles/d41586-026-00602-z |
| [4] | Gizmodo. Scientists Injected Stem Cells Into Fetuses to Treat a Birth Defect. Here’s What Happened. February 26, 2026.https://gizmodo.com/scientists-injected-stem-cells-into-fetuses-to-treat-a-birth-defect-heres-what-happened-2000727597 |
| [5] | Medical Xpress. First-ever in-utero stem cell therapy for fetal spina bifida repair shows safe results. February 26, 2026.https://medicalxpress.com/news/2026-02-utero-stem-cell-therapy-fetal.html |
| [6] | Medscape Medical News. First-in-Human Trial Supports Safety, Feasibility of Stem Cell Therapy in Prenatal Spina Bifida Repair. February 27, 2026.https://www.medscape.com/viewarticle/first-human-trial-supports-safety-feasibility-stem-cell-2026a1000667 |
| [7] | Medical Dialogues. In-Utero Stem Cell Therapy for Myelomeningocele Shows Early Safety and Feasibility: The Lancet. March 4, 2026.https://medicaldialogues.in/obstetrics-gynaecology/news/in-utero-stem-cell-therapy-for-myelomeningocele-shows-early-safety-and-feasibility-the-lancet-165830 |
| [8] | Knoepfler P. Early encouraging UC Davis trial data on cell therapy for spina bifida. The Niche. March 6, 2026.https://ipscell.com/2026/03/early-encouraging-uc-davis-trial-data-on-cell-therapy-for-spina-bifida/ |
| [9] | Progress Educational Trust. In utero stem cell therapy for spina bifida found safe in early trial. March 2026.https://www.progress.org.uk/in-utero-stem-cell-therapy-for-spina-bifida-found-safe-in-early-trial/ |
| [10] | UC Davis Health. World’s first stem cell treatment for spina bifida delivered during fetal surgery. October 2022.https://health.ucdavis.edu/news/headlines/worlds-first-stem-cell-treatment-for-spina-bifida-delivered-during-fetal-surgery–/2022/10 |
| [11] | Adzick NS, Thom EA, Spong CY, Farmer DL, et al. A Randomized Trial of Prenatal versus Postnatal Repair of Myelomeningocele. New England Journal of Medicine. 2011;364(11):993–1004.https://doi.org/10.1056/NEJMoa1014379 |
| [12] | Children’s Hospital of Philadelphia. Management of Myelomeningocele Study (MOMS). Center for Fetal Diagnosis and Treatment.https://www.chop.edu/centers-programs/center-fetal-diagnosis-and-treatment/management-myelomeningocele-study |
| [13] | Houtrow AJ, et al. Prenatal Repair of Myelomeningocele and School-Age Functional Outcomes (MOMS2). Pediatrics. January 2020.https://www.chop.edu/news/benefits-fetal-surgery-spina-bifida-persist-school-age-children |
| [14] | New Scientist. Stem cell patch reverses brain damage in fetuses with spina bifida. March 2026.https://lifeboat.com/blog/2026/03/stem-cell-patch-reverses-brain-damage-in-fetuses-with-spina-bifida |
| [15] | ClinicalTrials.gov. CuRe Trial: Cellular Therapy for In Utero Repair of Myelomeningocele. NCT04652908.https://clinicaltrials.gov/ct2/show/NCT04652908 |